 A03 Drugs for functional gastrointestinal disorders. The payee is not required to file an income tax return for the particular income. Failure to withhold the tax or in case of under withholding, the deficiency tax shall be collected from payor/withholding agent. National issues of the ATC classification may include additional codes not present in this list, which follows the WHO version. National issues of the ATC classification may include additional codes not present in this list, which follows the WHO version. The liability for payment of tax rests primarily on the payor as a withholding agent. Ĭodes for veterinary use ( ATCvet codes) can be created by placing the letter Q in front of the human ATC code: for example, QC05. 
The system provides a global standard for classifying medical substances and serves as a tool for drug utilization research. Subgroup C05 is part of the anatomical group C Cardiovascular system. Anatomical Therapeutic Chemical (ATC) classification system is maintained by the WHO Collaborating Centre for Drug Statistics Methodology in Oslo, Norway. The classification of a substance in the ATC/DDD system is therefore not a recommendation for use and it does not imply any judgements about efficacy or relative efficacy of drugs and groups of drugs.ATC code C05 Vasoprotectives is a therapeutic subgroup of the Anatomical Therapeutic Chemical Classification System, a system of alphanumeric codes developed by the World Health Organization (WHO) for the classification of drugs and other medical products. An important aim of drug utilization is to monitor rational as well as irrational drug use as an important step in improving the quality of drug use. The locations for the sonar drops are taken from the operators proposed procedure for clearing the site. It is essential that a tool for drug utilization monitoring and research is able to cover most medicines available on the market. 8 Registered Address 8A Zip Code 9 Registered Address 9A Zip Code 10 Foreign Address 10A Zip Code 10B ICR No. The EphMRA classification system is used worldwide by IMS (Intercontinental Medical Statistics) in producing marketing research statistics for the pharmaceutical industry. For this reason the ATC/DDD system by itself is not suitable for guiding decisions about reimbursement, pricing and therapeutic substitution. In some settings, and on the EphMRA website, the system is referred to as the ATC classification and this has caused confusion among users over the years. 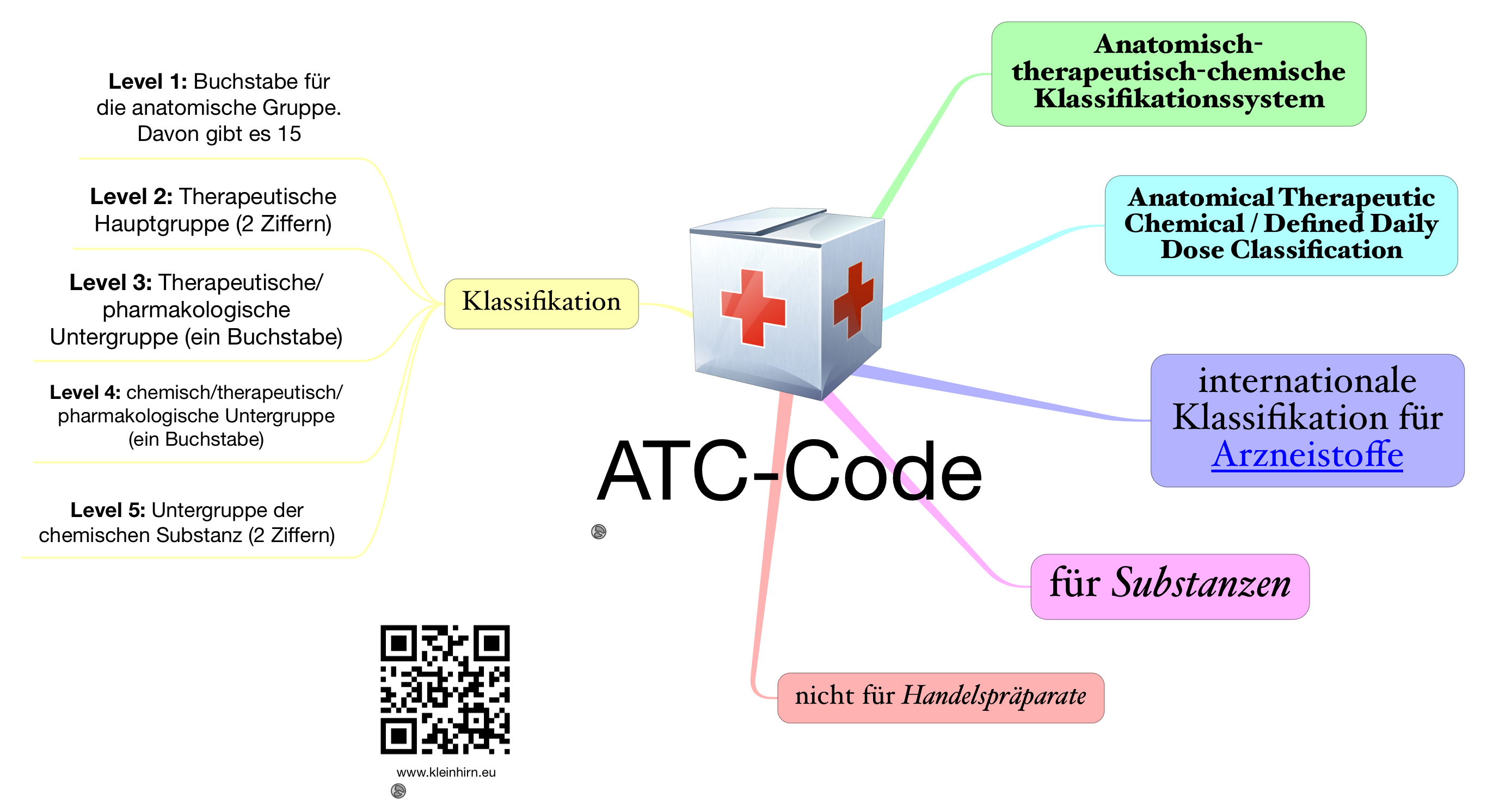
There is a strong reluctance to make changes to classifications or DDDs where such changes are requested for reasons not directly related to drug consumption studies. One component of this is the presentation and comparison of drug consumption statistics at international and other levels.Ī major aim of the Centre and Working Group is to maintain stable ATC codes and DDDs over time to allow trends in drug consumption to be studied without the complication of frequent changes to the system. 
The purpose of the ATC/DDD system is to serve as a tool for drug utilization monitoring and research in order to improve quality of drug use. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
